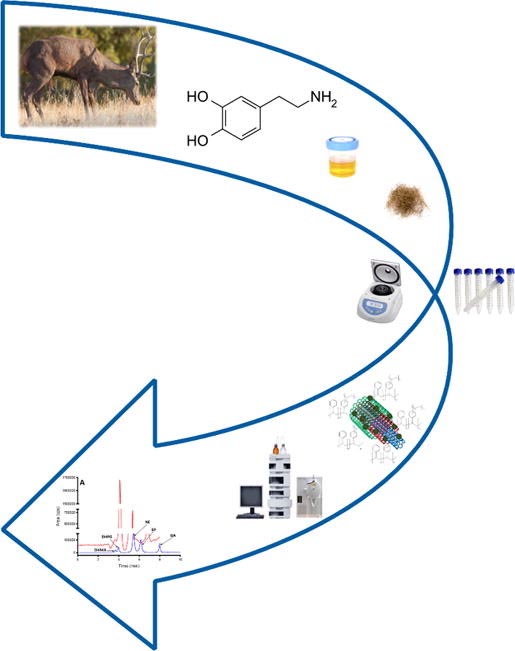
A novel analytical methodology for the determination and extraction of catecholamines (dopamine, epinephrine and norepinephrine) and their metabolites DL-3,4-dihydroxyphenyl glycol and DL-3,4-dihydroxymandelic acid by LC-MS is here developed and validated for application to human and animal urine and hair samples. The method is based on the preliminary extraction of analytes by a magnetic multi-walled carbon nanotube poly(styrene-co-divinylbenzene) composite. This is followed by a < 9 min chromatographic separation of the target compounds in an Onyx Monolithic C18 column using a mixture of 0.01% (v/v) heptafluorobutyric acid in water and methanol at 500 µL min-1 flow rate. Detection limits within range from 0.055 to 0.093 µg mL-1, and precision values of the response and retention times of analytes were > 90%. Accuracy values comprised the range 79.5–109.5% when the analytes were extracted from deer urine samples using the selected MMWCNT-poly(STY-DVB) sorbent. This methodology was applied to real red deer urine and hair samples, the resulting concentrations within range from 0.05 to 0.5 µg mL-1for norepinephrine and from 1.0 to 44.5 µg mL-1 for its metabolite 3,4-dihydroxyphenyl glycol. Analyses of red deer hair resulted in high amounts of 3,4-dihydroxyphenyl. informacion[at]ebd.csic.es: Murtada et al (2019) LC-MS determination of catecholamines and related metabolites in red deer urine and hair extracted using magnetic multi-walled carbon nanotube poly(styrene-co-divinylbenzene) composite. J Chromatogr B https://doi.org/10.1016/j.jchromb.2019.121878
https://www.sciencedirect.com/science/article/abs/pii/S157002321931150X